- Silvia Oliver-Mas, Jordi A. Matias-Guiu, Cristina Delgado-Alonso, et.al.
Abstract
Cognitive dysfunction is one of the most prevalent symptoms in patients with post-COVID condition (PCC) and episodic memory has been highlighted as one of the most impaired cognitive domains in these patients. However, few studies have specifically assessed episodic memory processes in these patients. This study aims to comprehensively investigate the memory function in patients with PCC. We conducted a cross-sectional study involving 157 patients with PCC and 74 healthy controls. Participants underwent a comprehensive neuropsychological assessment and memory assessment, including the Loewenstein-Acevedo Scale for Semantic Interference and Learning (LASSI-L), the Free and Cued Selective Reminding test (FCSRT), the Open-Trial Selective Reminding Test (OT-SRT), and a Mental Ability Questionnaire (FLEI). We compared groups and evaluated the correlation between episodic memory and neuropsychological and clinical assessments. Patients with PCC showed reduced performance on the LASSI-L, the FCSRT and the OT-SRT compared to controls. The memory scores showed positive moderate correlations with attention tests and positive low correlations with language, visuospatial or executive functions. Subjective memory complaints were related to poorer memory performance. LASSI-L was the test most associated with subjective memory complaints, whereas OT-SRT was the test less influenced by attention tests. The study found multiple memory processes impaired in patients with PCC, specifically in initial encoding, learning information acquisition and storage, and in retrieval, with only partial improvement with cues and recognition, and with significant susceptibility to the effects of retroactive semantic interference. These findings are relevant for characterising the cognitive deficits of patients with PCC and for designing interventional strategies.
Similar content being viewed by others
Long-term consequences of COVID-19 on sleep, mental health, fatigue, and cognition: a preliminary study
Article Open access01 May 2025

Evidence for a severe cognitive subgroup in a comprehensive neuropsychological Post-COVID-19 syndrome classification
Article Open access18 November 2025

Neurological post-COVID syndrome is associated with substantial impairment of verbal short-term and working memory
Article Open access11 January 2025
Explore related subjects
Discover the latest articles, books and news in related subjects, suggested using machine learning.
Introduction
Post-COVID condition (PCC) involves individuals with a history of probable or confirmed SARS-CoV-2 infection, whose symptoms persist more than three months since the infection [1]. Different symptoms have been described, and the most common symptoms are fatigue, cognitive dysfunction, and shortness of breath. Females have a higher likelihood of presenting PCC than males [2]. PCC mostly affects young people of working age, in their daily living activities, their quality of life, and employment status [3, 4]. Specifically, cognitive deficits have a negative impact on the work capacity of patients with PCC and are associated with functional impairment [5, 6]. Research has consistently demonstrated cognitive dysfunction in patients with PCC. Cognitive impairment may involve different domains [7], but the most common deficits are in attention, processing speed [8,9,10] and executive function [11, 12].
Memory complaints are also frequent in PCC, and episodic memory is one of the main cognitive domains impaired according to previous research [13, 14]. Episodic memory allows individuals to store and consciously remember personal experiences [15]. Episodic memory involves three main steps: encoding information, storage/consolidation, and retrieval [16]. In brief, encoding is the process of converting information into a format that can be stored in either long-term or short-term memory [17] and attention deficits can affect negatively [18]. Then, the consolidation process stores information for later use and the retrieval process recovers the stored information [19].
Neuropsychological assessment of learning new information processes is important for understanding the basis of episodic memory. Previous studies in other neurological conditions have used memory instruments such as the Free and Cued Selective Reminding Test (FCSRT) [20], Loewenstein-Acevedo Scales for Semantic Interference and Learning (LASSI-L) [21], and the Open Trial Selective Reminding Test (OT-SRT) [22] to assess specific processes such as interference susceptibility, learning efficiency and cued response [23,24,25,26,27,28].
In PCC, episodic memory has been assessed with several tests in the context of general neuropsychological batteries [29]. Specifically, some of these studies have found episodic memory impairment [30, 31] while others have found no impairment [32, 33]. The differences in results could be due to variations in sample size, patients’ clinical characteristics, or the heterogeneity of memory assessments. Therefore, it is crucial to thoroughly analyse the memory processes in PCC using appropriate tools to perform an accurate evaluation of the patients. A proper assessment of memory processes includes measuring learning, response to cues, and proactive and retroactive interference, and the use of several memory tests could reduce the particular issues of each task.
However, to our knowledge, the neuropsychological processes underlying episodic memory impairment in PCC are unclear. We hypothesized that, due to the pathophysiology of the cognitive dysfunction of PCC in which several mechanisms are involved [34] there will be several cognitive processes involved in memory deficits. Because memory issues are frequent in PCC and are one of the most disabling symptoms [8, 35], in this study we aimed to comprehensively investigate the memory function in these patients. We aimed to examine the main memory processes (encoding, consolidation and retrieval), their relationship with other cognitive functions (especially attention and processing speed) and clinical scales. Additionally, we explored the link between objective memory performance and subjective cognitive complaints. We used three objective memory tests: the FCSRT [20], LASSI-L [21], OT-SRT [22], and a subjective cognitive questionnaire (FLEI) [36], in order to examine memory under different paradigms.
Materials and methods
Study design and participants
This is a cross-sectional study that involved 231 participants: 157 patients with PCC, and 74 healthy controls (HC). Both groups were similar in age, sex, and years of education (Supplementary Table S1).
All the participants in the study were native Spanish speakers to ensure valid results in Spanish verbal memory assessment, as language plays a crucial role in cognitive assessment [37]. Inclusion criteria for PCC patients were: (1) Diagnosis of SARS-CoV-2 infection with positive RT-PCR at least three months before being included in the study; (2) Diagnosis of PCC according to WHO criteria; [1] (3) Subjective cognitive complaints since the SARS-CoV-2 infection. Exclusion criteria were the following: (1) The existence of cognitive dysfunction or cognitive complaints prior to SARS-CoV-2 infection; (2) Previous or current diagnosis of neurological disorder (e.g. stroke or brain injury), psychiatric disease (e.g. schizophrenia) or infection in the central nervous system with cognitive impairment; (3) History of alcohol consumption, drugs or other toxics that impact cognitive ability; (4) Sensory or motor deficits that may hinder the memory assessment. For the HC group, exclusion criteria were: (1) subjective cognitive complaints or cognitive impairment; (2) medical history of neurological disorders associated with cognitive impairment (e.g. traumatic brain injury); (3) present or previous confirmed psychiatric disorder with a potential impact on cognitive functions (e.g. depression or schizophrenia); (4) History of substance abuse; (5) Cognitive impairment due to uncontrolled medical conditions or drugs at the moment of the assessment; (6) Sensory disorder which can possibly affect the results of cognitive assessment.
Comprehensive memory assessment
Participants underwent a comprehensive memory assessment which included the LASSI-L [21], the FCSRT [20], and a subgroup was also assessed with the OT-SRT [22]. Episodic memory assessments were conducted in different sessions.
The Loewenstein-Acevedo scales for semantic interference and learning (LASSI-L)
The LASSI-L is a cognitive stress paradigm that uses controlled learning and cued recall to maximize storage and retrieval of the 15 written words [21]. The test consists of two lists of words (List A and List B), composed by 15 words each, corresponding to three semantic categories (fruits, musical instruments, and articles of clothing), with five words in each category. The LASSI-L evaluates semantic interference, which is the difficulty of learning or remembering information due to the interaction of learning material with past or new memories [26] and for this reason it could be more ecological.
The test is administered according to the following procedure. To start, the first list of 15 words from three categories (List A) is presented on cards by the examiner and the patient has to read out the words, afterward, the examiner asks the participant to recall all words (Free recall A, FRA1) (this score is a measure of initial encoding). Secondly, the patient is asked again to recall the words, but this time a semantic cue is provided for the three categories (e.g., “Now I want you to tell me all the words from the list that are fruits”) (Cued Recall A, CRA1). Then, List A is presented again, and a cued recall is performed again (CRA2) (maximum storage). Immediately, List B is presented following the same procedure as List A. The examiner presents to the participant the 15 different words (with same categories: fruits, musical instruments, and articles of clothing) and then the examinee is asked for the Free Recall B 1 (FRB 1), Cued Recall B 1 (CRB1), and Cue Recall B 2 (CRB2) following the same procedure as List A (FRB1 and CRB1 are associated with proactive semantic interference, and CRB2 refers to the recovery from proactive semantic interference); 5) Thereafter, participant is asked to free recall the words of List A (Short- delay free recall, SdFRA), and then the participant is asked to recall the words when prompted with cues from List A (Short delay cued recall (SdCRA). SdFRA and SdCRA are associated with retroactive interference effects. After 20 min, the participant is asked for the delayed recall (DR) of both lists. For each trial, the number of words that were remembered correctly were recorded, as well as semantic intrusions that occurred. Time allowed for each task is 60 s for free recall, 20 s for cued recalls, and 90 s for the DR.
The free and cued selective reminding test (FCSRT)
The FCSRT measures episodic memory by testing the participant’s ability to learn and recall a list of 16 written words [20]. Each word is presented with a semantic cue to control for the memory encoding process (e.g., “raven” with its semantic category, “bird”). Participants are required to recall words in three trials, during 90 s each trial. Each trial consists of free recall of the list of words, and then, the examiner provides the semantic cues of those words from the list that the participant did not recall, therefore, also uses a control learning paradigm.
Finally, free and cued delayed recall are evaluated after 30 min. Hence, the FCSRT consisted of various scores, including Trial 1 Free Recall (which is the free recall of the list, a measure of initial encoding), Total Free Recall (which is the sum of free recall during the learning trials, and it refers to learning), Total Recall (which is the sum of free recall and cued recall during the learning trials, a measure of storage), Delayed Free Recall, Delayed Total Recall (which is the sum of delayed free recall and delayed cued recall).
The open trial selective reminding test (OT-SRT)
The Open Trial Selective Reminding Test (OT-SRT) was administered to a subgroup of 55 patients with PCC and 55 HC, similar in age, sex and education were recruited for specific comparisons (Supplementary Table S1).
The Open Trial Selective Reminding Test (OT-SRT) is an effective tool for assessing the learning process across the number of trials required for the subject to learn new words [38], which is useful to assesses whether memory impairment is associated with acquisition, storage, or retrieval difficulties [39]. OT-SRT consists of reading to the participant a list of 10 words that are semantically related (i.e., clothing) in a maximum of 15 trials. Immediately after that, the examinee is asked to recall as many items as possible. In successive trials, the examiner will repeat to the participant the words that were not remembered from the previous trial. Participants continue the list learning until a criterion of complete recall (10 words) on two consecutive trials is achieved. Then, 30 min later participant is required to recall the list of words and a recognition task is performed.
Neuropsychological and clinical assessment
In addition to the episodic memory assessment, patients with PCC underwent a comprehensive neuropsychological and clinical evaluation, conducted by a trained neuropsychologist. The protocol included the following tests: Forward and backward Digit Span, Corsi block tapping test (forward and backward), Symbol Digit Modalities Test (SDMT), Boston Naming Test (BNT), Judgment of Line Orientation (JLO), Rey-Osterrieth Complex Figure (ROCF) (time, copy and recall at 3, 30 min, and recognition), verbal fluencies (animals and words beginning with “p”, “m” and “r” in 1 min each one), Stroop Color Word Interference Test (W: words reading; C: colors naming; and WC: interference words-colors), and the Visual Object and Space Perception Battery (VOSP). These tests were co-normed and validated in several neurological disorders in our setting [40, 41].
The clinical assessment included the following scales: State-Trait Anxiety Inventory (STAI) [42], Beck Depression Inventory (BDI-II) [43], Pittsburgh Sleep Quality Index (PSQI) [44], Brief Smell Identification Test (BSIT) [45], and the Modified Fatigue Impact Scale (MFIS) [46]. Based on previous studies, the following cut-offs were applied: STAI-S ≥ 40 was considered clinically significant anxiety [47] BDI-II ≥ 19 was regarded as moderate or severe depression [48]; BSIT ≤ 8 was categorized as having abnormal olfaction; PSQI > 5 defined poor sleep quality [49] and MFIS ≥ 38 was considered as having fatigue [50].
Mental ability questionnaire (FLEI)
The FLEI questionnaire is a subjective questionnaire that encompasses 35 questions about subjective cognitive complaints associated with memory (FLEI-Memory), attention (FLEI-Attention), and executive functions (FLEI-Executive Functions). A total score (FLEI-Total) is also obtained as the sum of FLEI-Memory, FLEI-Attention and FLEI-Executive Function [36]. Low raw scores or high percentile ranks indicate high subjective cognitive ability, while high raw scores or low percentile ranks indicate low subjective cognitive ability. Scores can be divided into 3 subgroups: “below-average level” when the percentile is lower than 16, “average level” when the percentile ranks between 16 and 84, and “above-average level” when the percentile is higher than 84 [51].
Statistical analyses
Statistical analysis was carried out with SPSS Statistics 22.0. Comparisons between groups were performed with T-test or χ2 test when appropriate, and statistical significance was set at p < 0.05. The effect size was estimated using Cohen’s d for the comparison of two means, considering the effect to be small (d = 0.2), moderate (d = 0.5) or large (d = 0.8). Correlation analyses were performed with Pearson’s coefficient (r). Bonferroni correction was applied for correlation analyses, correlations with cognition were corrected by the number of cognitive test at p < 0.0017. Similarly, correlations with clinical aspects were corrected at p < 0.01, and correlations with FLEI test and subscores were corrected at p < 0.0125. Correlations were regarded as low (rho < 0.30), moderate (rho = 0.30–0.49), or high (rho > 0.49).
Likewise, the percentage of impairment in FCSRT was calculated with normative data available in the Neuronorma Project [40]. Impairment was set at two cut-off scores, scaled score of ≤ 7 (percentile ≤ 16) and scale score of ≤ 5 (percentile ≤ 5).
Ethics statement
The present study was approved by the ethics committee from Hospital Clínico San Carlos (reference 21/062-E) and all participants gave written informed consent.
Results
Characteristics of PCC patients
PCC patients were recruited at 16.96 ± 7.98 months after acute infection. The mean age of PCC and HC was 45.80 ± 9.06 years and 44.72 ± 8.45 years respectively, (p = 0.386). Regarding sex, there were 115 (73.24%) women in PCC group and 46 (62.16%) women in the HC group (p = 0.087). Also, PCC patients had a mean of 15.57 ± 3.07 years of education compared to HC (mean of 15.73 ± 3.29) (p = 0.714). Premorbid risk factors are described in Supplementary Table S1. Regarding the clinical profile, 50.6% of PCC patients showed anxiety symptoms, 30.3% reported depressive symptoms, 89% sleep quality dysfunction, 23.9% presented with olfaction problems and 89% reported fatigue.
Moreover, concerning subjective cognitive complaints (FLEI questionnaire), 84.3% of PCC participants revealed a below-average percentile, showing low subjective cognitive ability, while 15% of participants performed at an average level, and 0.7% of participants performed above-average level. PCC participants also performed below average in FLEI subscores, specifically 87.6% in FLEI-Memory, 86.3% in FLEI-Attention, and 63.4% in FLEI-Executive Functions.
LASSI-L performance in PCC compared to HC
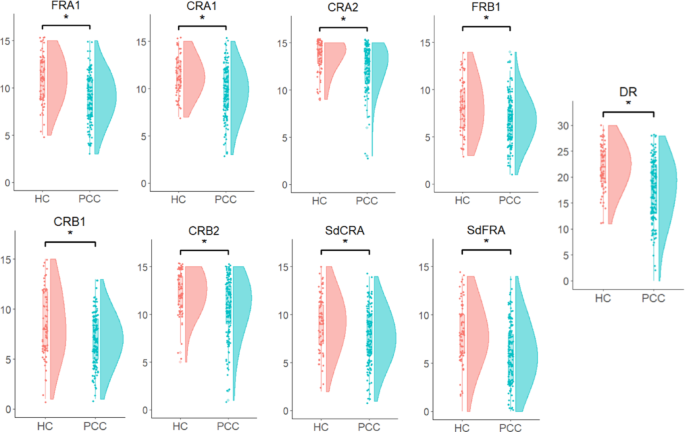
There were statistically significant differences in all LASSI-L measures between PCC and HC (p < 0.001) (Table 1; Fig. 1). Effect sizes were moderate for FRA1 (d = 0.762), CRA1 (d = 0.763), CRA2 (d = 0.508), FRB1 (d = 0.528), CRB1 (d = 0.548), CRB2 (d = 0.563), SdFRA (d = 0.646), and SdCRA (d = 0.526). Moreover, effect size was large for DR (d = 0.879).Table 1 LASSI-L performance in PCC compared to HC
Additionally, we investigated whether these differences remained significant after controlling for the learning ability. Therefore, we performed the same analyses with FRA1 as a covariate and the results remained significant (Supplementary Table S2).
LASSI-L associations with neuropsychological, clinical and FLEI performance in PCC patients
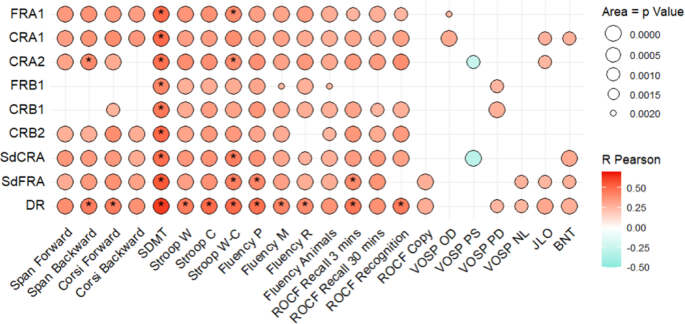
LASSI-L subscores showed larger correlations with attention, processing speed and episodic memory tests, and were scarcely associated with language, visuoperceptual or visuospatial performance (Fig. 2). No significant correlations were found between LASSI-L subscores and clinical aspects (Supplementary Table S3).
Scarce and low correlations were found between LASSI-L with some subscores of clinical tests (Supplementary Table S3).
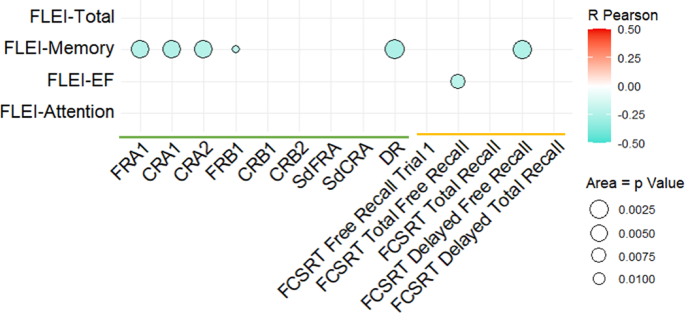
Regarding associations with subjective cognitive complaints, several LASSI-L subscores (FRA1, CRA1, CRA2, FRB1 and DR) were inversely associated with the FLEI-Memory subscore (p < 0.0125). That is, the more subjective memory complaints, the poorer performance in LASSI-L (Fig. 3, Supplementary Table S3).
FCSRT performance and associations with neuropsychological, clinical and FLEI performance in PCC patients
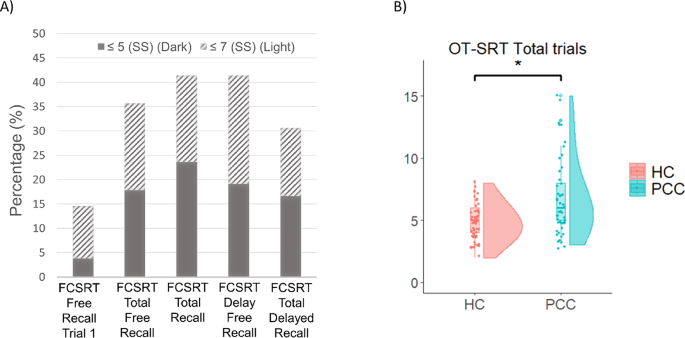
PCC patients presented with impairment in all FCSRT subscores, more accentuated in total recall (41.4%) and delayed free recall (41.4%) (Table 2; Fig. 4.A).Table 2 Percentage of FCSRT impairment in PCC
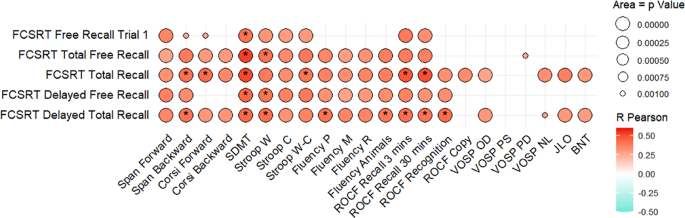
FCSRT subscores showed larger and significant correlations with attention, processing speed and episodic memory tests (p < 0.001). These correlations were generally moderate. Correlations with verbal fluency, language, visuoperceptual or visuospatial performance were weaker (Fig. 5, Supplementary Table S3).
Scarce and low correlations were found between FCSRT with some subscores of clinical tests (Supplementary Table S3). Small correlations were found between FCSRT scores (total free recall, total recall, and total delay recall) and BDI-II (Supplementary Table S3). Finally, significant correlations were found between FCSRT Delayed Free Recall and FLEI-Memory and between FCSRT total free recall and FLEI-Executive Functions (Fig. 3, Supplementary Table S3).
OT-SRT performance and associations with neuropsychological, clinical and FLEI performance in PCC patients
The OT-SRT was administered to a subgroup of 55 patients PCC and 55 HC, similar in age, sex and education. PCC subgroup had a mean age of 48.02 ± 8.08 years and HC had 47.69 ± 9.09 years (p = 0.842). Regarding sex, 80% of the subgroup of PCC patients were women and 76.36% of the HC patients were women (p = 0.644). Moreover, PCC patients had a mean of 16.98 ± 3.36 years of education and HC had a mean of 17.11 ± 4.11 years of education (p = 0.861) (Supplementary Table S1).
PCC patients required significantly more trials to learn the 10 words than HC (p < 0.001) (Fig. 4. B). Similarly, PCC revealed significant differences in total trials and recall at 30 min compared to HC (p < 0.001) with moderate effect size (d = 0.742 and d = 0.778, respectively) and small effect size in recognition false positives (d = 0.416). No significant differences were found in recognition (Table 3).Table 3 OT-SRT performance in PCC compared to HC
No significant correlations were found between OT-SRT total trials or delay recall with cognitive tests, clinical assessment or FLEI (Supplementary Table S3).
Correlations between episodic memory tests
CRA2 and DR showed high correlations with the FCSRT scores measuring learning and recall (rho = 0.549–0.632). FRA1 and FCSRT (free recall trial 1) showed a moderate (rho = 0.322) correlation. Regarding the OT-SRT, total trials showed moderate and negative correlations with some LASSI-L scores (FRA1 rho = −0.436, CRA1 rho = −0.494, CRB1 = −0.437, CRB2 = −0.414, SdFRA = −0.436, and SdCRA = −0.468) and with delayed free recall (rho = −0.560) and delayed total recall of the FCSRT (rho=−0.493). OT-SRT (delayed recall) showed a large correlation with FRA1 (rho = 0.522) and moderate correlations with CRA1 (rho = 0.388), CRA2 (rho = 0.300), CRB2 (rho = 0.321), SdFRA (rho = 0.331), SdCRA (rho = 0.447) and FCSRT (free recall trial 1) (rho = 0.420). All the correlations are shown in Supplementary Table S4-S5.
Discussion
In this study, the episodic memory performance of individuals with PCC and cognitive complaints was assessed using three neuropsychological tests based on different paradigms. PCC patients revealed reduced performance in all three administered memory tests compared to controls. The use of three different episodic memory tests along with a comprehensive neuropsychological assessment and clinical evaluation of other important symptoms in PCC permit a broad and thorough overview of the memory dysfunction in this disorder.
During the LASSI-L, we observed that patients with PCC performed worse than controls in all the scores. Similarly, in the FCSRT, the percentage of impairment in considering both cutoffs (scaled-score 5 and scaled-score 7) was greater than expected in almost all the scores. Regarding the OT-SRT, we also found a lower performance in PCC in the main scores. Overall, this suggests that, independently of the cognitive test administered, PCC group showed a lower performance in episodic memory compared with controls or normative data. The moderate effect sizes suggest a potential meaningful association between variables, which may have clinical relevance.
The effect size for the LASSI-L was greater for the delayed (free) recall. Similarly, for the FCSRT, the percentage of impairment was greater for the equivalent score (delayed free recall), and for the OT-SRT, the recall at 30 min was lower than in controls. Importantly, delayed recall in both LASSI-L and FCSRT were the scores more associated with subjective memory complaints, confirming the relevance of these scores from the patient’s perspective. This highlights the importance of including a subjective cognitive test in the clinical evaluation of PCC, as they capture the real-life cognitive challenges that patients face, even when objective deficits are subtle or heterogeneous [35].
Regarding the analysis of the specific memory processes impaired, it is worth noting that during the LASSI-L, the effect sizes were greater in the initial encoding (FRA1 and CRA1) than in the maximum storage (CRA2). However, when adjusting for the initial encoding, the other scores remained statistically significant. This suggests that, although the impairment in the initial encoding exists, this does not explain all the memory deficits. In this regard, the initial encoding in the FCSRT (Free Recall Trial 1) showed a low percentage of impairment, and the first trial of the OT-SRT did not show significant differences between patients and controls. This is consistent with the finding that the correlation between initial encoding and attentional tests was higher for the LASSI-L than for the other tests. The differences in the learning strategy of each test, the characteristics, and the number of items to be remembered could probably explain these findings.
Notably, we found difficulties in the learning process and maximum storage in the three memory tests. In the FCSRT, the percentage of impairment reached 23.6–41.4% of patients, and in the OT-SRT, the patients needed more trials to complete the recall of the 10 target words. Similarly, the performance was lower in CRA2 in the LASSI-L. Thus, the learning difficulties are present despite the two different procedures used in the tests (i.e. focus the attention of examinees towards unrecalled words in FCSRT and OT-SRT in successive trials vs. only two presentations of all the words list in LASSI-L).
Another interesting finding is regarding the benefit of semantic cues during recall. In the FCSRT, the percentage of patients with a scaled-score ≤ 7 at the delayed recall improved from 41.4% without cues to 30.6% with semantic cues. In the OT-SRT, there were statistically significant differences in the recall at 30 min and the recognition errors, but not in recognition success (although there was a trend towards statistical significance). Overall, this suggests that patients improve with cues and recognition, but partially. This finding was also observed in a previous study [52].
One of the advantages of the LASSI-L paradigm is the assessment of learning under the effects of proactive and retroactive semantic interference. The effect size was higher in retroactive interference, which means PCC are more vulnerable to the effects of retroactive semantic interference, which may be due to difficulties in episodic memory and executive function [8, 53]. The prominent involvement of retroactive semantic interference was also found in multiple sclerosis (MS) [54], which shares cognitive similarities with PCC including attention and processing speed deficits [55]. Interestingly, it differs from prodromal Alzheimer’s disease, in which there is a selective failure in the recovery from the effects of proactive semantic interference [25, 26]. It is also remarkable that the subscores that measure retroactive interference have a high correlation with the Stroop Test and SDMT, two tests associated with focused attention, inhibition abilities, working memory, and processing speed. It reinforces the idea that retroactive semantic interference is related to attention and executive function difficulties.
Our findings have important practical implications for the neuropsychological assessment of memory in PCC and the selection of the optimal cognitive test. In this regard, the LASSI-L showed the highest correlation with the subjective cognitive complaints questionnaire. This is consistent with the fact that LASSI-L is examining learning and recall during the effects of proactive and retroactive interference, and such interference in memory is likely to occur in daily living. This observation confirms the ecological value of the LASSI-L. However, the LASSI-L and FCSRT are both correlated with several attention, processing speed and executive tests. This suggests that the performance of both tests is partially explained by other cognitive functions, which should be taken into account in the clinical interpretation. In this regard, the use of a less challenging procedure for learning (e.g. more trials, fewer words) in the OT-SRT seems to be a good approach to reduce the influence of attention in the assessment of episodic memory.
These findings are consistent with previous literature that identified episodic memory impairment as an important deficit in patients with PCC [13, 14]. Also, the use of multiple memory paradigms including LASSI-L, FCSRT and OT-SRT allowed to capture subtle impairments that may not be detected using more traditional tools. Additionally, the results regarding retroactive interference vulnerability are consistent with prior findings in multiple sclerosis [54] and reinforce the hypothesis that executive dysfunction may contribute to the memory profile in PCC.
Moreover, the present findings could have clinical implications. First, they highlight the importance of conducting memory assessments in patients with PCC, particularly using paradigms that consider interference effects and differentiate between encoding, storage, and retrieval. Given that memory deficits and memory complaints are highly prevalent and subjectively distressing in this population [13, 14, 56], neuropsychological evaluation can help validate patients’ experiences and guide cognitive rehabilitation strategies. Additionally, the partial benefit observed in PCC with semantic cues and recognition task suggests that memory deficits in PCC could be mitigated through compensatory strategies.
Our study has some limitations. First, we evaluated the memory performance in the PCC as a group. A detailed analysis of the individual memory profiles could be also interesting in future studies, especially in combination with neuroimaging or other pathophysiological biomarkers of the disease. Second, OT-SRT was only applied to a subgroup of patients, however, this subgroup showed no significant differences compared to the group assessed with LASSI-L and FCSRT. Additionally, it is important to note that our findings apply to patients who have symptoms after the infection. Individuals who are infected but do not report symptoms would probably have a different outcome [14, 57, 58].
In conclusion, our study found multiple memory processes impaired in patients with PCC. Specifically, we observed difficulties in initial encoding, a delay in learning and information acquisition and storage, and impairment in retrieval with only partial improvement with cues and recognition and with greater effects from retroactive semantic interference. These findings are relevant for the characterization of the cognitive characteristics of patients with PCC, for the choice of the cognitive tests for episodic memory assessment, and for the design of interventional strategies.
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
- Soriano JB, Murthy S, Marshall JC, Relan P, Diaz JV (2022) A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect Dis 22:e102–e107Article CAS PubMed Google Scholar
- Ganesh R, Grach SL, Ghosh AK et al (2022) The female-predominant persistent immune dysregulation of the post-COVID syndrome. Mayo Clin Proc 97:454–464Article CAS PubMed Google Scholar
- Gualano MR, Rossi MF, Borrelli I, Santoro PE, Amantea C, Daniele A, Tumminello A, Moscato U (2022) Returning to work and the impact of post COVID-19 condition: a systematic review. Work 73:405–413PubMed Google Scholar
- de Oliveira Almeida K, Nogueira Alves IG, de Queiroz RS, de Castro MR, Gomes VA, Santos Fontoura FC, Brites C, Neto MG (2023) A systematic review on physical function, activities of daily living and health-related quality of life in COVID-19 survivors. Chronic Illn 19:279–303Article PubMed Google Scholar
- Delgado-Alonso C, Cuevas C, Oliver-Mas S, Díez-Cirarda M, Delgado-Álvarez A, Gil-Moreno MJ, Matías-Guiu J, Matias-Guiu JA (2022) Fatigue and cognitive dysfunction are associated with occupational status in post-COVID syndrome. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph192013368Article PubMed PubMed Central Google Scholar
- Jaywant A, Gunning FM, Oberlin LE, Santillana M, Ognyanova K, Druckman JN, Baum MA, Lazer D, Perlis RH (2024) Cognitive symptoms of post–COVID-19 condition and daily functioning. JAMA Netw Open 7:e2356098–e2356098Article Google Scholar
- Ceban F, Ling S, Lui LMW et al (2022) Fatigue and cognitive impairment in post-COVID-19 Syndrome: a systematic review and meta-analysis. Brain Behav Immun 101:93–135Article CAS PubMed Google Scholar
- Delgado-Alonso C, Valles-Salgado M, Delgado-Álvarez A et al (2022) Cognitive dysfunction associated with COVID-19: a comprehensive neuropsychological study. J Psychiatr Res 150:40–46Article PubMed PubMed Central Google Scholar
- Herrera E, Pérez-Sánchez MdelC, San Miguel-Abella R et al (2023) Cognitive impairment in young adults with post COVID-19 syndrome. Sci Rep. https://doi.org/10.1038/s41598-023-32939-0Article PubMed PubMed Central Google Scholar
- Krishnan K, Miller AK, Reiter K, Bonner-Jackson A (2022) Neurocognitive profiles in patients with persisting cognitive symptoms associated with COVID-19. Arch Clin Neuropsychol 37:729–737Article PubMed Google Scholar
- Bertuccelli M, Ciringione L, Rubega M, Bisiacchi P, Masiero S, Del Felice A (2022) Cognitive impairment in people with previous COVID-19 infection: a scoping review. Cortex. https://doi.org/10.1016/j.cortex.2022.06.002Article PubMed PubMed Central Google Scholar
- Miskowiak KW, Johnsen S, Sattler SM, Nielsen S, Kunalan K, Rungby J, Lapperre T, Porsberg CM (2021) Cognitive impairments four months after COVID-19 hospital discharge: pattern, severity and association with illness variables. Eur Neuropsychopharmacol 46:39–48Article CAS PubMed PubMed Central Google Scholar
- Aiello EN, Fiabane E, Manera MR, Radici A, Grossi F, Ottonello M, Vassallo C, Pain D, Pistarini C (2022) Episodic long-term memory in post-infectious SARS-CoV-2 patients. Neurol Sci 43:785–788Article PubMed Google Scholar
- Zhao S, Shibata K, Hellyer PJ, Trender W, Manohar S, Hampshire A, Husain M (2022) Rapid vigilance and episodic memory decrements in COVID-19 survivors. Brain Commun. https://doi.org/10.1093/braincomms/fcab295Article PubMed PubMed Central Google Scholar
- Tulving E (1993) What is episodic memory? Curr Dir Psychol Sci 2:67–70Article Google Scholar
- DeLuca J, Christodoulou C, Diamond BJ, Rosenstein ED, Kramer N, Ricker JH, Natelson BH (2004) The nature of memory impairment in chronic fatigue syndrome. Rehabil Psychol 49:62–70Article Google Scholar
- Craik FIM (2007) Encoding: a cognitive perspective. In: Roediger 3rd HL et al., editors. Science of memory: concepts. NewYork: Oxford University Press. p. 129–135.
- Mangels JA, Picton TW, Craik FIM (2001) Attention and successful episodic encoding: an event-related potential study. Cogn Brain Res. https://doi.org/10.1016/S0926-6410(00)00066-5. https://doi.org/Article Google Scholar
- Howe ML (1988) 2. Measuring Memory Development in Adulthood: A Model-Based Approach to Disentangling Storage-Retrieval Contributions
- Buschke H (1984) Cued recall in amnesia. J Clin Neuropsychol 6:433–440Article CAS PubMed Google Scholar
- Crocco E, Curiel RE, Acevedo A, Czaja SJ, Loewenstein DA (2014) An evaluation of deficits in semantic cueing and proactive and retroactive interference as early features of Alzheimer’s disease. Am J Geriatr Psychiatry 22:889–897Article PubMed Google Scholar
- Chiaravalloti ND, Balzano J, Moore NB, DeLuca J (2009) The open-trial selective reminding test (OT-SRT) as a tool for the assessment of learning and memory. Clin Neuropsychol 23:231–254Article PubMed Google Scholar
- Gainotti G, Quaranta D, Vita MG, Marra C (2014) Neuropsychological predictors of conversion from mild cognitive impairment to Alzheimer’s disease. J Alzheimers Dis 38:481–495Article CAS PubMed Google Scholar
- Teichmann M, Epelbaum S, Samri D, Levy Nogueira M, Michon A, Hampel H, Lamari F, Dubois B (2017) Free and cued selective reminding test – accuracy for the differential diagnosis of Alzheimer’s and neurodegenerative diseases: a large-scale biomarker-characterized monocenter cohort study (ClinAD). Alzheimers Dement 13:913–923Article PubMed Google Scholar
- Matias-Guiu JA, Cabrera-Martín MN, Curiel RE, Valles-Salgado M, Rognoni T, Moreno-Ramos T, Carreras JL, Loewenstein DA, Matías-Guiu J (2018) Comparison between FCSRT and LASSI-L to detect early stage Alzheimer’s disease. J Alzheimers Dis 61:103–111Article CAS PubMed PubMed Central Google Scholar
- Loewenstein DA, Curiel RE, Duara R, Buschke H (2018) Novel cognitive paradigms for the detection of memory impairment in preclinical Alzheimer’s disease. Assessment 25:348–359Article PubMed Google Scholar
- DeLuca J, Barbieri-Berger S, Johnson SK (1994) The nature of memory impairments in multiple sclerosis: acquisition versus retrieval. J Clin Exp Neuropsychol 16:183–189Article CAS PubMed Google Scholar
- DeLuca J, Schultheis MT, Madigan NK, Christodoulou C, Averill A (2000) Acquisition versus retrieval deficits in traumatic brain injury: implications for memory rehabilitation. Arch Phys Med Rehabil 81:1327–1333Article CAS PubMed Google Scholar
- Knapp SAB, Austin DS, Aita SL, Caron JE, Owen T, Borgogna NC, Del Bene VA, Roth RM, Milberg WP, Hill BD (2024) Neurocognitive and psychiatric outcomes associated with postacute COVID-19 infection without severe medical complication: a meta-analysis. J Neurol Neurosurg Psychiatry. https://doi.org/10.1136/jnnp-2024-333950Article PubMed Google Scholar
- Ariza M, Cano N, Segura B et al (2022) Neuropsychological impairment in post-COVID condition individuals with and without cognitive complaints. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2022.1029842Article PubMed PubMed Central Google Scholar
- Akıncı B, Oğul ÖE, Hanoğlu L, Kulaç B, Ören D, Ulu O, Basançelebi B (2023) Evaluation of cognitive functions in adult individuals with COVID-19. Neurol Sci 44:793–802Article PubMed Google Scholar
- Dondaine T, Ruthmann F, Vuotto F, Carton L, Gelé P, Faure K, Deplanque D, Bordet R (2022) Long-term cognitive impairments following COVID-19: a possible impact of hypoxia. J Neurol 269:3982–3989Article CAS PubMed PubMed Central Google Scholar
- Hadad R, Khoury J, Stanger C, Fisher T, Schneer S, Ben-Hayun R, Possin K, Valcour V, Aharon-Peretz J, Adir Y (2022) Cognitive dysfunction following COVID-19 infection. J Neurovirol. https://doi.org/10.1007/s13365-022-01079-yArticle PubMed PubMed Central Google Scholar
- Monje M, Iwasaki A (2022) The neurobiology of long COVID. Neuron 110:3484–3496Article CAS PubMed PubMed Central Google Scholar
- Delgado-Alonso C, Díez-Cirarda M, Pagán J et al (2025) Unraveling brain fog in post-COVID syndrome: relationship between subjective cognitive complaints and cognitive function, fatigue, and neuropsychiatric symptoms. Eur J Neurol 32:e16084Article PubMed Google Scholar
- Beblo T, Kunz M, Brokate B, Scheurich A, Weber B, Albert A, Richter P, Lautenbacher S (2010) Entwicklung eines Fragebogens Zur subjektiven einschätzung der geistigen leistungsfähigkeit (FLei) Bei patienten Mit Psychischen störungen. Z Fur Neuropsychologie 21:143–151Article Google Scholar
- Stålhammar J, Hellström P, Eckerström C, Wallin A (2022) Neuropsychological test performance among native and non-native swedes: second language effects. Arch Clin Neuropsychol 37:826–838Article PubMed Google Scholar
- Buschke H (1973) Selective reminding for analysis of memory and learning. J Verbal Learn Verbal Behav 12:543–550Article Google Scholar
- DeLuca J, Gaudino EA, Diamond BJ, Christodoulou C, Engel RA (1998) Acquisition and storage deficits in multiple sclerosis. J Clin Exp Neuropsychol 20:376–390Article CAS PubMed Google Scholar
- Peña-Casanova J, Blesa R, Aguilar M et al (2009) Spanish multicenter normative studies (NEURONORMA project): methods and sample characteristics. Arch Clin Neuropsychol. https://doi.org/10.1093/arclin/acp027Article PubMed Google Scholar
- Peña-Casanova J, Casals-Coll M, Quintana M, Sánchez-Benavides G, Rognoni T, Calvo L, Palomo R, Aranciva F, Tamayo F, Manero RM (2012) Spanish normative studies in a young adult population (NEURONORMA young adults project): methods and characteristics of the sample. Neurología (English Edition). https://doi.org/10.1016/j.nrleng.2011.12.008Article Google Scholar
- Spielberger C, Gorsuch R, Lushene R, Vagg PR, Jacobs G (1983) Manual for the State-Trait Anxiety Inventory (Form Y1 – Y2)
- Beck AT, Steer RA, Brown G (1996) Beck depression Inventory–II. https://doi.org/10.1037/t00742-000. Psychol Assess. https://doi.org/
- Buysse DJ, Reynolds Ill CF, Monk TH, Berman SR, Kupfer DJ (1989) The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. https://doi.org/10.1016/0165-1781(89)90047-4Article PubMed Google Scholar
- Doty RL, Avron;, Lee M, WW (1996) Development of the 12-Item Cross-Cultural Smell Identification Test (CC-SIT). https://doi.org/10.1097/00005537-199603000-00021
- Kos D, Kerckhofs E, Carrea I, Verza R, Ramos M, Jansa J (2005) Evaluation of the modified fatigue impact scale in four different European countries. Mult Scler 11:76–80Article CAS PubMed Google Scholar
- Spielberger CD, Gonzalez-Reigosa F, Martinez-Urrutia A, Natalicio LFS, Natalicio DS (2017) The State-Trait anxiety inventory. Revista Interamericana De Psicología/Interamerican J Psychol. https://doi.org/10.30849/rip/ijp.v5i3Article Google Scholar
- Beck, Steer RA, Carbin MC (1988) Psychometric properties of the Beck depression inventory: twenty-five years of evaluation. Clin Psychol Rev. https://doi.org/10.1016/0272-7358(88)90050-5. https://doi.org/Article Google Scholar
- Buysse DJ, Hall ML, Patrick;, Strollo J, Kamarck TW, Owens J, Lee L, Reis SE, Matthews KA (2008) Epworth sleepiness scale (ESS), and clinical/ polysomnographic measures in a community sample. J Clin Sleep Med. https://doi.org/10.5664/jcsm.27351Article PubMed PubMed Central Google Scholar
- Strober LB, Bruce JM, Arnett PA et al (2020) Tired of not knowing what that fatigue score means? Normative data of the modified fatigue impact scale (MFIS). Mult Scler Relat Disord. https://doi.org/10.1016/j.msard.2020.102576Article PubMed PubMed Central Google Scholar
- Beblo T, Kunz M, Albert A, Aschenbrenner S, Debelak R (2011) Manual mental ability questionnaire test label FLEI
- Llana T, Mendez M, Zorzo C, Fidalgo C, Juan MC, Mendez-Lopez M (2022) Anosmia in COVID-19 could be associated with long-term deficits in the consolidation of procedural and verbal declarative memories. Front Neurosci. https://doi.org/10.3389/fnins.2022.1082811Article PubMed PubMed Central Google Scholar
- Möller M, Borg K, Janson C, Lerm M, Normark J, Niward K (2023) Cognitive dysfunction in post-COVID-19 condition: mechanisms, management, and rehabilitation. J Intern Med 294:563–581Article PubMed Google Scholar
- Matias-Guiu JA, Cortés-Martínez A, Curiel RE, Delgado-Álvarez A, Fernández-Oliveira A, Pytel V, Montero P, Moreno-Ramos T, Loewenstein DA, Matías-Guiu J (2020) Memory impairment in relapsing-remitting multiple sclerosis using a challenging semantic interference task. Front Neurol. https://doi.org/10.3389/fneur.2020.00309Article PubMed PubMed Central Google Scholar
- Delgado-Alonso C, Delgado-Alvarez A, Díez-Cirarda M et al (2024) Cognitive profile in multiple sclerosis and post-COVID condition: a comparative study using a unified taxonomy. Sci Rep. https://doi.org/10.1038/s41598-024-60368-0Article PubMed PubMed Central Google Scholar
- Delgado-Alonso C, Díez-Cirarda M, Pagán J et al (2023) Unraveling brain fog in post-COVID syndrome: relationship between subjective cognitive complaints and cognitive function, fatigue, and neuropsychiatric symptoms. Eur J Neurol. https://doi.org/10.1111/ene.16084Article PubMed PubMed Central Google Scholar
- Hampshire A, Azor A, Atchison C et al (2024) Cognition and memory after Covid-19 in a large community sample. N Engl J Med 390:806–818Article CAS PubMed PubMed Central Google Scholar
- Serrano del Pueblo VM, Serrano-Heras G, Romero Sánchez CM et al (2024) Brain and cognitive changes in patients with long COVID compared with infection-recovered control subjects. Brain. https://doi.org/10.1093/brain/awae101/7638850
