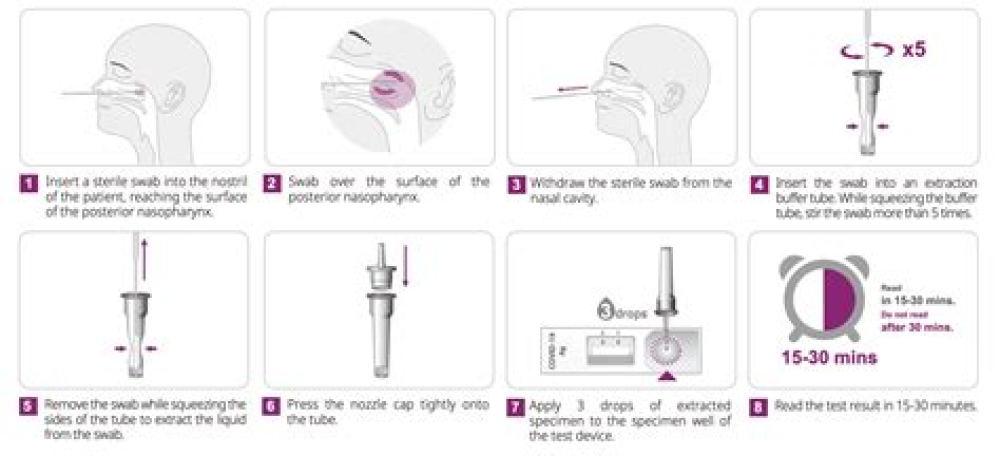
Authors: CDC
Summary of Recent Changes
Updates as of May 13, 2021
- Updated guidance based on new published studies on antigen test performance.
- Clarification about which NAATs should be used for confirmatory testing.
- Considerations for people who have had previous SARS-CoV-2 infections and those who have been fully vaccinated.
- Two new antigen testing algorithms, one for congregate living settings, and one for community settings.
- Updates to testing suggestions for fully vaccinated, asymptomatic people.
View Previous Updates
Key Points
- This interim guidance is intended for healthcare providers who order antigen tests, receive antigen test results, or perform point-of-care testing, as well as for laboratory professionals who perform antigen testing in a laboratory setting or at the point of care and report those results.
- The purpose of this interim technical guidance is to support effective clinical and public health use of antigen tests for different testing situations.
- This guidance applies to all clinical and consumer uses of antigen tests and is not specific to any particular age group.
- This guidance incorporates considerations for fully vaccinated people and should be used in conjunction with CDC’s Interim Public Health Recommendations for Fully Vaccinated People.
For More Information: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-guidelines.html